Thyroid Drugs Being Recalled After Supplier Fails Inspection
Some batches of thyroid medications are being recalled, after a Chinese supplier failed an FDA inspection.
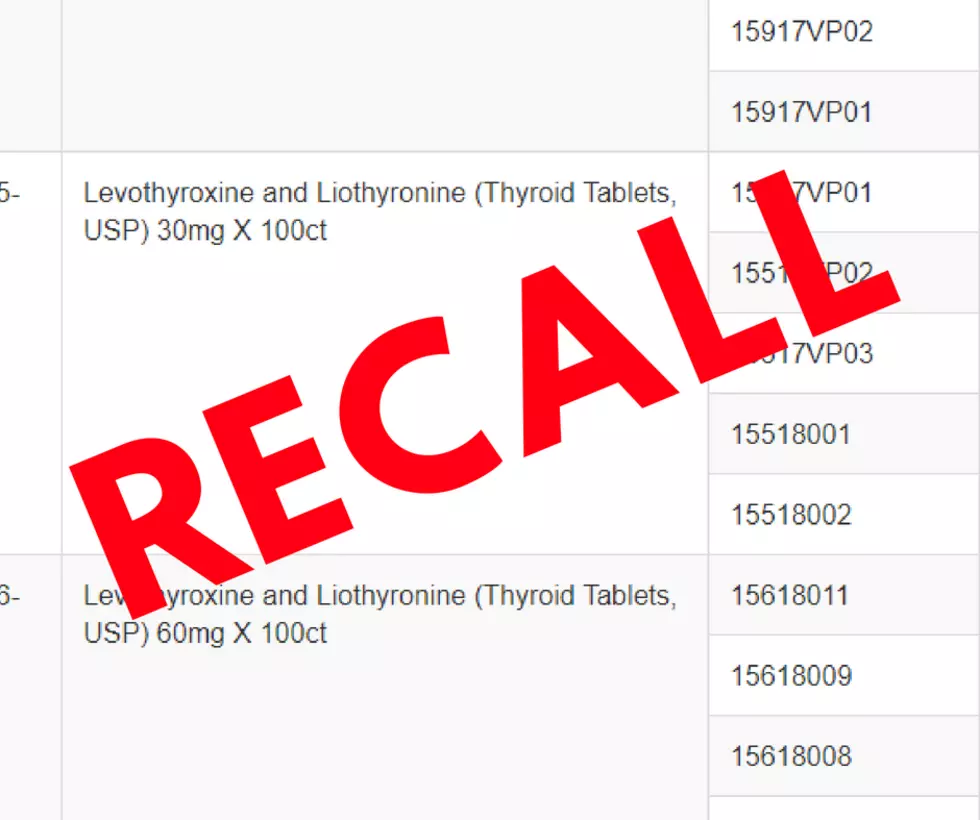
Westminster Pharmaceuticals, LLC is voluntarily recalling all lots of levothyroxine and liothyronine in strengths of 15 mg, 30 mg, 60 mg, 90 mg, & 120 mg. These tablets were sold at the wholesale level. The Chinese supplier is responsible for the active pharmaceutical ingredient used to manufacture the pills that were manufactured and distributed by Westminster Pharmaceuticals.
The thryoid medications, levothyroxine and liothyronine, are used to treat patients who suffer from hypothyroidism, and are used to replace or supplement the body's own thyroid hormone.
"Because these products may be used in the treatment of serious medical conditions, patients taking the recalled medicines should continue taking their medicine until they have a replacement product," according to FDA.gov.
Westminster is contacting all of its wholesale accounts "to immediately discontinue distribution of the product being recalled." They have not received any reports of adverse events related to this product. Consumers that have these products which are being recalled should not discontinue use before contacting their physician for further guidance.
Patients can also contact Westminster Pharmaceuticals with questions at 888-354-9939.
The full list of recalled medications is available on the FDA's website.
More From Lite 98.7









